Files
Download Chapter (814 KB)
Description
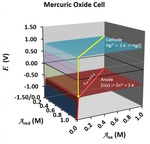
Two characteristics of batteries, their delivery of nearly constant voltage and their rapid failure, are explained through a visual examination of the Nernst equation. Two Galvanic cells are described in detail: (1) a wet cell involving iron and copper salts and (2) a mercury oxide dry cell. A complete description of the wet cell requires a three-dimensional Nernst surface because the potential is a function of two variables: the activities of both the oxidized and reduced forms in each redox couple. Dry cell potentials, which utilize solid or pure liquid species, are functions of only one variable and can be described by a pair of traces in a traditional plot. Plots of the Nernst potential are relatively flat for most activities, but they exhibit bluffs and cliffs under extreme conditions. The flat plateaus are responsible for the fairly constant voltage that batteries deliver; the bluffs and cliffs explain why batteries fail so quickly as they wear down. This chapter, an expansion of ideas introduced in Chapter 3.1, focuses on a familiar real-world application.
Publication Date
1-2022
Document Type
Book
Rights
© 2022 Garon C. Smith, Md Mainul Hossain, and Patrick MacCarthy
Creative Commons License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
Disciplines
Chemistry
Recommended Citation
Smith, Garon C.; Hossain, Md Mainul; and MacCarthy, Patrick, "Chapter 3.2: Why Batteries Deliver a Fairly Constant Voltage Until They Suddenly Die: An Application of Nernst Topo Surfaces" (2022). Water Topos: A 3-D Trend Surface Approach to Viewing and Teaching Aqueous Equilibrium Chemistry. 9.
https://scholarworks.umt.edu/topos/9
Comments
This document is the Accepted Manuscript version of a Published Work that appeared in final form in the Journal of Chemical Education, copyright © American Chemical Society and the Division of Chemical Education, Inc., after peer review and technical editing by the publisher. Access the final edited and published work.